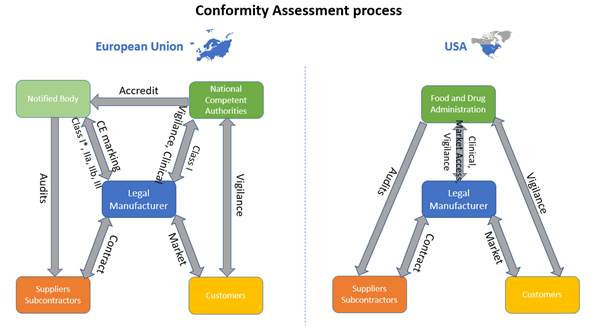
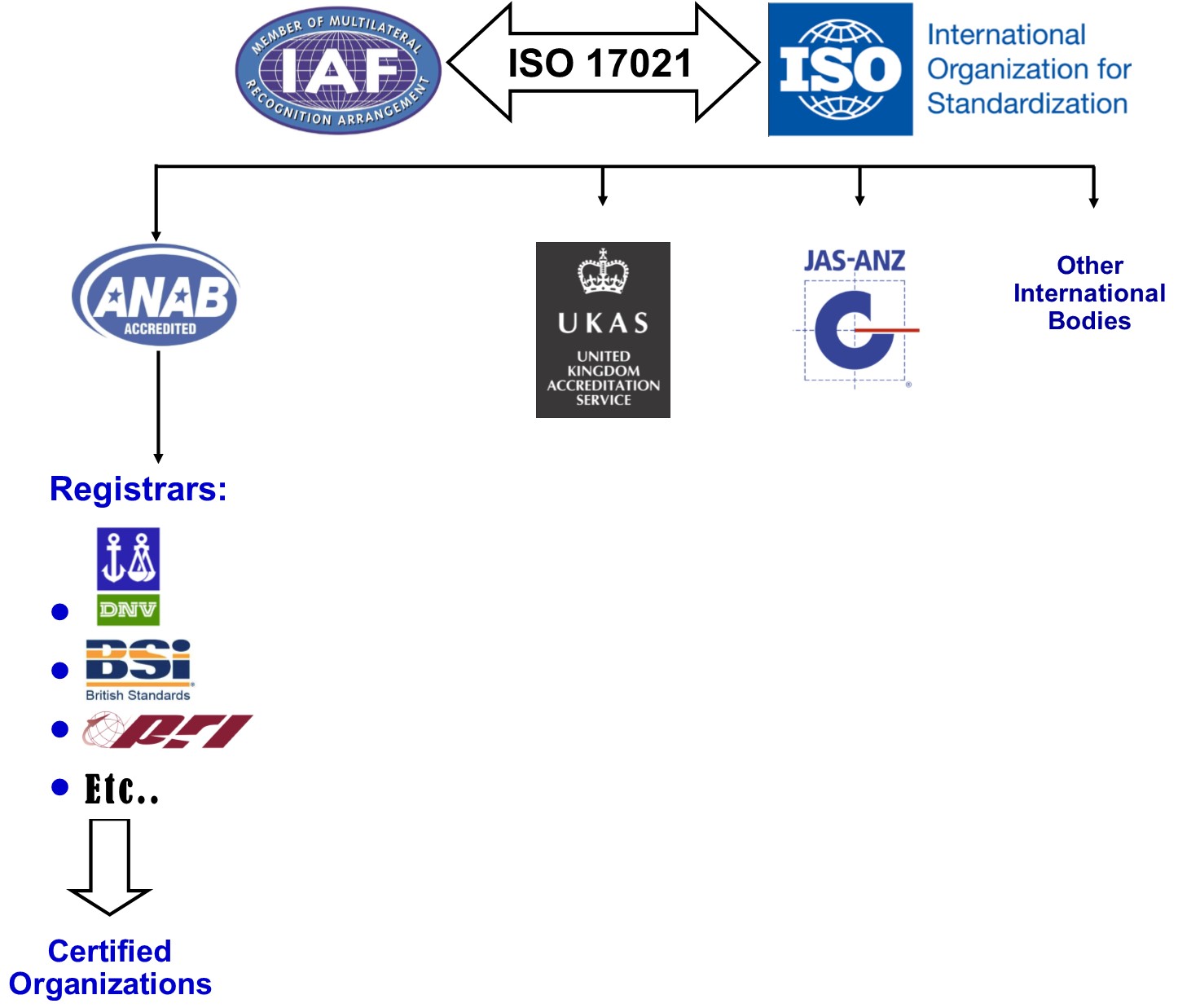
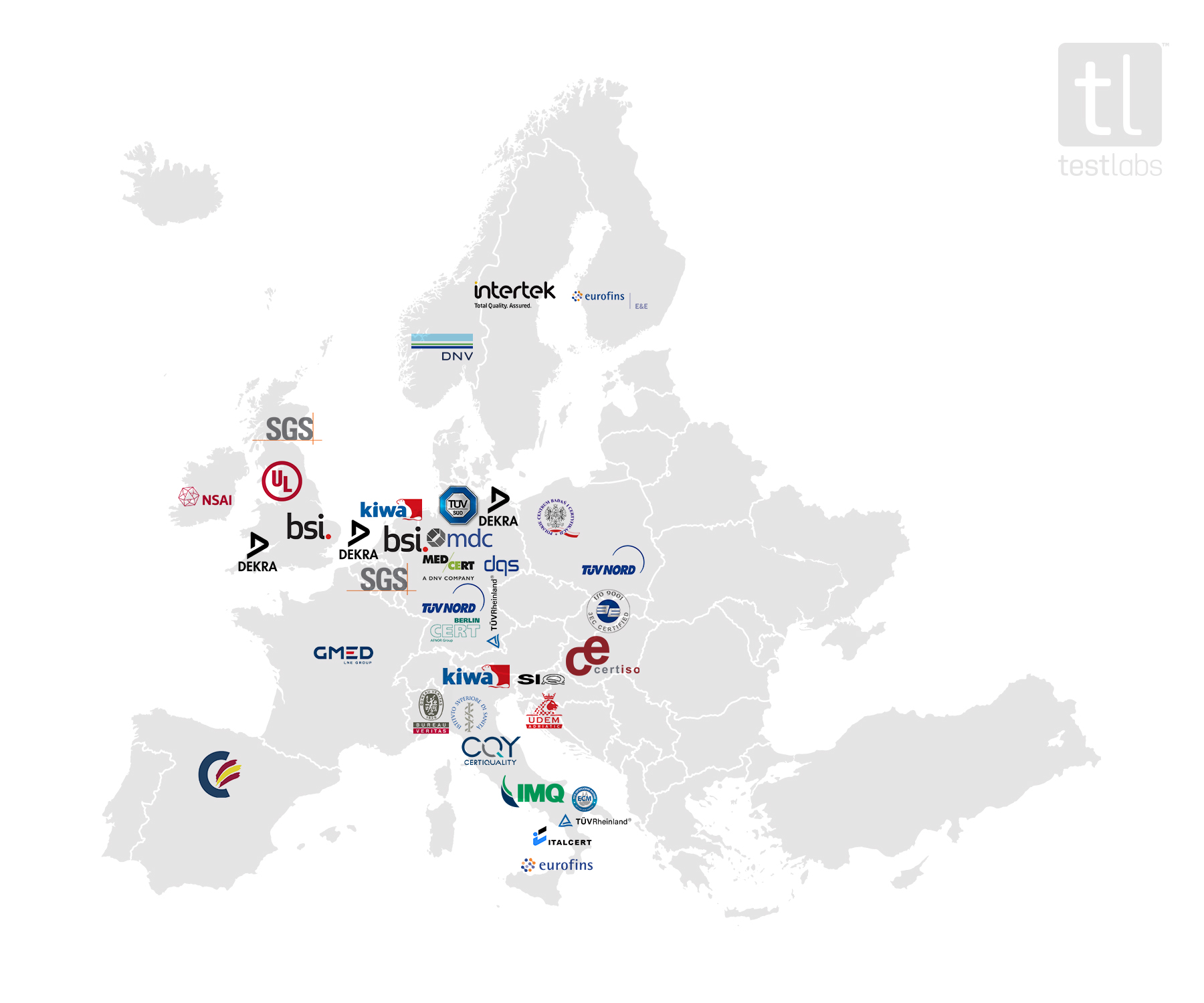
Availability and capacity of notified bodies to carry out conformity assessments for COVID-19 related medical devices and in vit

ECM Notified Body accredited for CE certification activities under Directive 2014/53/EU (RED) - Ente Certificazione Macchine